|
What is SnapPath®?
BioMarker Strategies developed the proprietary SnapPath® Cancer Diagnostics System* to guide targeted drug development and treatment selection for patients with solid tumor malignancies.
SnapPath® is the only diagnostics system able to generate purified populations of live solid tumor cells from fresh, unfixed samples in a rapid and standardized manner. SnapPath® preserves the molecular integrity of these live cells for ex vivo exposure to targeted therapies and combinations on the SnapPath® System. This enables generation of a new class of much more predictive biomarker tests. We call these new biomarker tests PathMAP® Functional Signaling Profiles.
The dynamic information available only from live cells is required to understand the acquired resistance that too often develops in the treatment of cancer. This understanding is necessary for accurate prediction of individual response to targeted treatment of solid tumors.
Currently available biomarker tests for solid tumors primarily rely on dead, fixed tissue samples. These tests are useful in identifying relevant mutations and suggesting general therapeutic approach, but they are not useful for understanding the mechanisms of acquired resistance, and they do not accurately predict individual response to targeted treatment.
The PathMAP® Functional Signaling Profiles enabled by the SnapPath® System provide direct information about the dynamic response of live solid tumor cell signaling pathways to targeted treatments – and are useful in identifying and understanding mechanisms of acquired resistance.
PathMAP® Functional Signaling Profiles are highly predictive of individual response to targeted treatment.
How does it work?
SnapPath® is a highly customizable automated, fluidics-based Cancer Diagnostic System. It consists of a compact bench-top instrument and a disposable cartridge that allow for the collecting, processing and testing of fresh, unfixed solid tumor tissue samples.
SnapPath® rapidly processes live solid tumor cells from a fine needle aspiration biopsy, surgical excision or other fresh, unfixed tumor tissue sample. SnapPath® is compatible with any solid tumor for which fresh tissue samples are available. The process begins with the loading of fresh tissue samples onto the disposable cartridge, which is then snapped into place on the SnapPath® instrument.
SnapPath® Automated Processing takes as little as thirty minutes and involves the following steps:
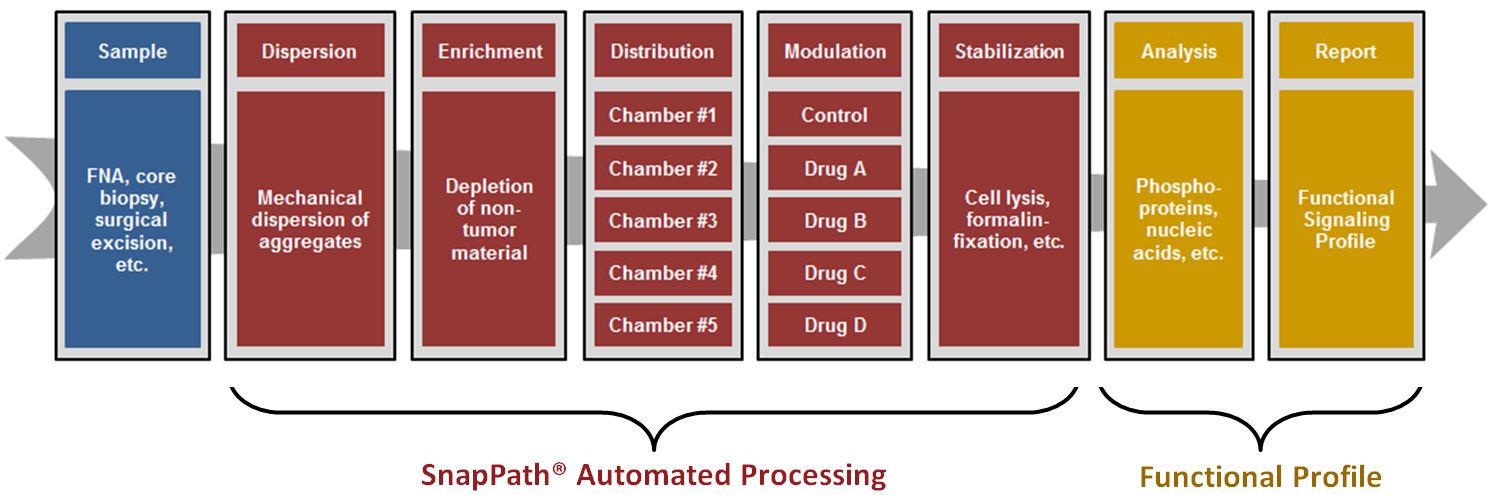
Dispersion: In the dispersion step, the fresh solid tumor tissue samples are mechanically disaggregated on the SnapPath® System into smaller groupings of living solid tumor cells more suitable for stimulation, inhibition and molecular analysis.
Enrichment: Following disaggregation, non-tumor cells such as red blood cells and white blood cells are removed from the sample on the System using antibody-coated magnetic beads, thereby enriching the tumor sample population to facilitate downstream molecular analysis.
Distribution: Following enrichment, the live sample is distributed via automated fluidics into multiple test chambers for ex vivo modulation. The number of test wells—up to five—is dependent on the input, selected analytes and preferred downstream analytical method.
Modulation: Following distribution into test wells, the live solid tumor cells are exposed to targeted therapies (inhibitors) or various stimulators (growth factors). By modulating live cells in this manner, the SnapPath® platform evokes phosphoprotein-based PathMAP® Functional Signaling Profiles that are subsequently compared against the unstimulated (“basal”) sample. A portion of the sample is reserved for traditional biomarker analysis.
Stabilization: Following modulation, samples are stabilized for off-platform extraction and phosphoprotein analysis. The SnapPath® System is agnostic to the type of analytical technology used.
What are the key advantages of the SnapPath® System?
The evolution of personalized medicine is changing the way oncologists diagnose and treat cancer. Approximately 50% of compounds currently in development are molecularly targeted drugs. The pharmaceutical industry is also rapidly moving beyond the development of single-target agents guided by single biomarker tests, to the development of drugs and drug combinations with complex mechanisms of action that will benefit from pathway-based diagnostics derived from an increasing understanding of the complex signal transduction network of living cells.
The PathMAP® Functional Signaling Profiles enabled by the SnapPath® System provide users with snapshots of the dynamic nature of cellular processes. BioMarker Strategies believes that the SnapPath® System has the potential to usher in a new era where fresh, unfixed solid tumor tissue can be routinely and rapidly analyzed rather than relying solely on dead, fixed tissue samples that provide limited insight into the biological complexity of solid tumor cells.
*For Research Use Only (RUO). Not currently intended for use in diagnostic procedures.
|






